Solid Phase Oligonucleotide Synthesis Is Not Obsolete - It Is Ascending
Why phosphoramidite‑based, flow‑enabled solid‑phase synthesis remains the manufacturing backbone of therapeutic oligonucleotides
Publication Metrics
Read Time: 7 Min
Word Count: 1,290

AI Transparency
Abstract:
The last five years have produced meaningful progress in enzymatic oligonucleotide synthesis, liquid‑phase strategies, stereocontrolled phosphorus chemistry, and process intensification. At the same time, regulators have sharpened expectations around impurity characterization, batch definition, and integrated control strategies for synthetic oligonucleotides. The net result is not platform replacement, but convergence. Solid-phase phosphoramidite synthesis (SPOS) is being modernized, especially through flow-enabled execution, while emerging approaches occupy complementary niches.
This paper shows, from a chemistry and CMC perspective, that SPOS remains the only platform that simultaneously delivers chemical generality, mechanistically understood impurity formation, orthogonal analytical tractability, and regulatory-mature lifecycle continuity for therapeutic oligonucleotides. The newest literature and regulatory commentary reinforce a future where the manufacturing ‘reference chemistry’ persists, while execution evolves [13, 11, 10].
1. What “Standard” Means in Regulated Oligonucleotide Manufacturing
In regulated pharmaceutical manufacturing, “standard” does not mean “best for every use case.” It means a platform is:
- Inspectable
- Comparable across lifecycle changes
- Chemically general across therapeutically relevant modifications
- Analytically resolvable with established orthogonal methods and impurity taxonomies [10, 18, 4].
Regulatory discussions in the last few years increasingly emphasize integrated control strategies, impurity assessment, and clear batch definition for synthetic oligonucleotides—topics that fit directly with SPOS unit operations and decades of process understanding [10, 18, 11].
2. Therapeutic Oligonucleotides Are Chemical Objects, Not Just Sequences
Therapeutic oligonucleotides derive key clinical properties from chemical architecture (sugar/backbone modifications, end‑caps, conjugations, and stereochemical distributions, rather than sequence alone [4, 3]. This favors platforms that can reliably incorporate a wide range of modifications under controlled conditions, which remains a defining strength of phosphoramidite SPOS [13, 11].
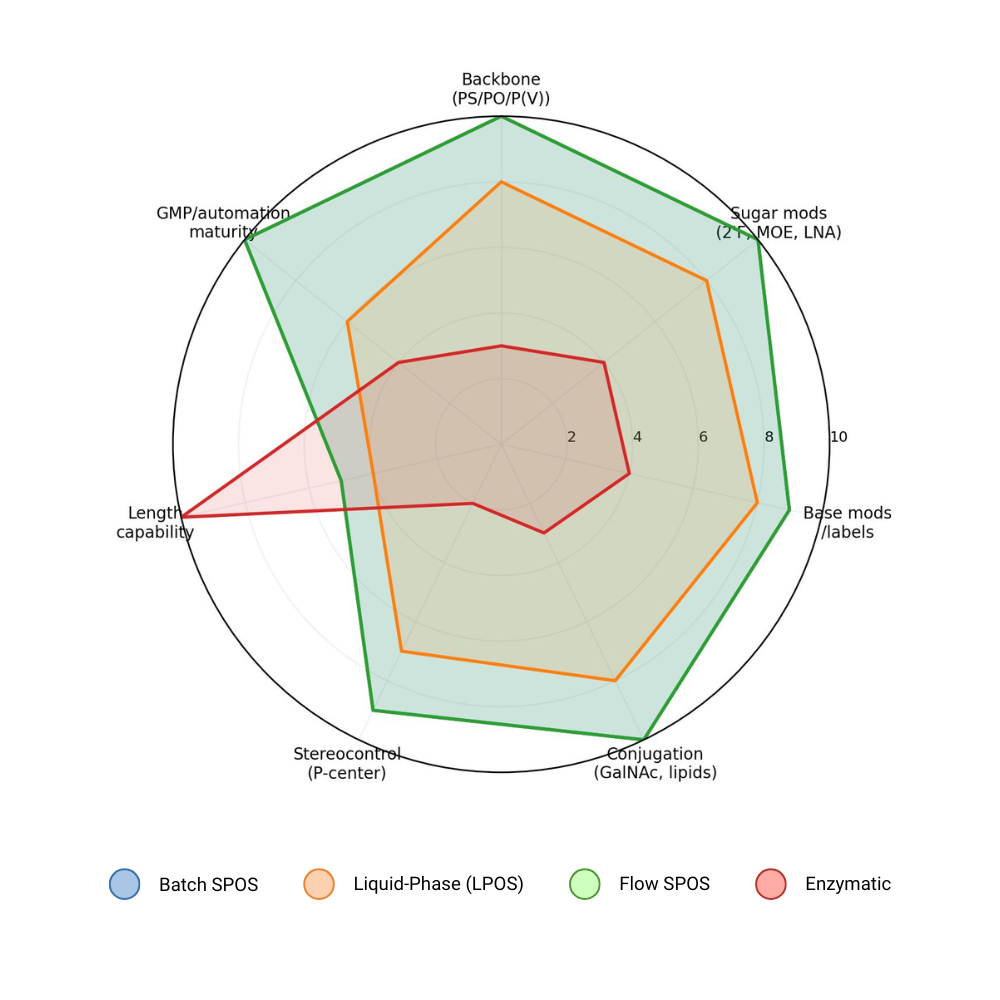
Figure 1.
Relative chemical generality of oligonucleotide synthesis platforms.
3. Impurity Science: Predictability is an Advantage
SPOS impurity mechanisms include:
- Deletions (n–1, n–2)
- Depurination-linked cleavage pathways
- Capping-associated base modifications
- Oxidation and sulfurization variants
These are chemically interpretable and analytically addressable within established control strategies [10, 18].
Regulatory discussions reinforce that the industry is judged not by the absence of impurities, but by the ability to detect, control, trend, and justify them [18, 10]. Emerging enzymatic workflows can reduce solvent burden and avoid certain SPOS‑specific side reactions, but they introduce their own platform‑specific impurity sets and risk classes, remain early in their regulatory learning curve [14, 17, 12].
4. Flow Chemistry: The Modern Form of Solid‑Phase Synthesis
Most criticism of SPOS has historically targeted batch execution, not phosphoramidite chemistry itself. Modern process intensification—especially flow‑through solid‑phase architectures—improves mass transfer and residence‑time control, reduces exposure to aggressive reagents, and increases uniformity and reproducibility [5, 6]. The result is reduced solvent intensity and improved execution while preserving the same core reaction network and comparability logic [13, 11].
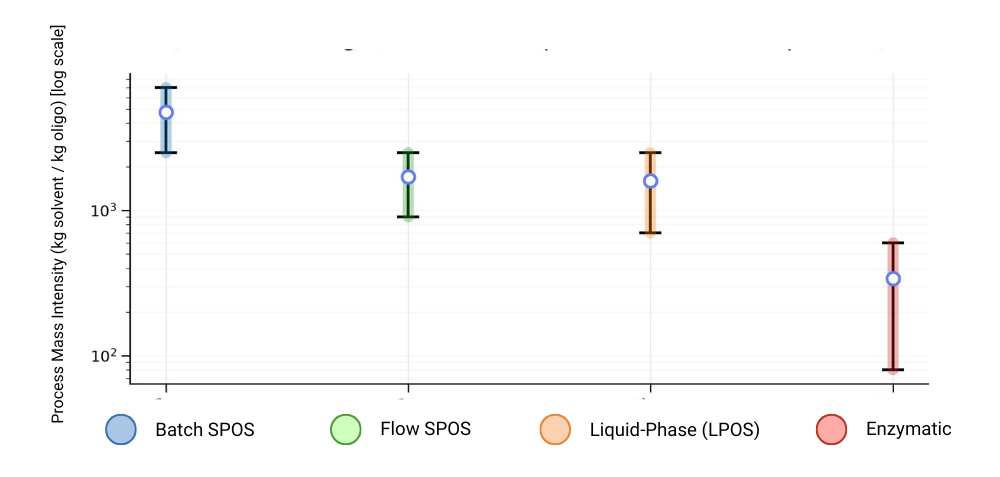
Figure 2. Representative PMI ranges by platform (illustrative ranges; actual PMI is process‑ and scale‑dependent).
5. Regulatory Maturity Follows Lifecycle Continuity, Not Novelty
Regulators favor platforms that produce consistent quality attributes across time and change, especially where impurity profiles are well understood and analytical methods are mature. Industry regulatory analyses and regulator communications emphasize integrated control strategies, clear batch definition (including pooling/splitting considerations), and robust impurity characterization [10, 18].
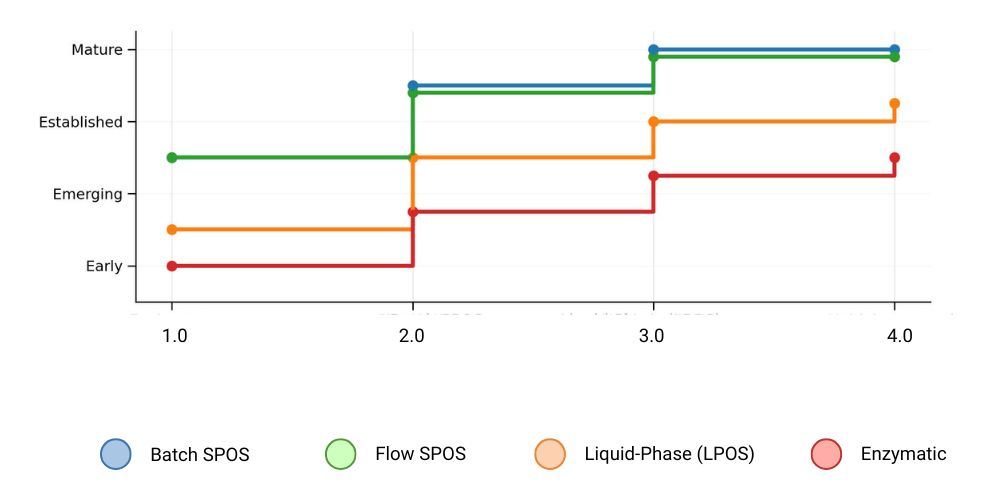
Figure 3. Conceptual regulatory / quality‑system maturity by platform (qualitative lifecycle framing; not an approval count).
6. Recent Advances (2020–2025): Evolution, Not Replacement
Between 2020 and 2025, oligonucleotide manufacturing advanced significantly due to:
- Sustainability pressures
- Expanding therapeutic complexity
- The need for scalable production
Key developments include:
- Flow-enabled solid-phase phosphoramidite synthesis reducing solvent use and improving mass transfer while preserving established reaction chemistry and impurity profiles [11, 13].
- Advances in phosphorus chemistry and stereocontrol, expanding the range of accessible therapeutic structures [13, 15]
- Progress in enzymatic oligonucleotide synthesis under aqueous conditions, including controlled incorporation and template-independent approaches [17, 14, 12]
Despite this progress, enzymatic approaches remain constrained in chemical generality and regulatory precedent. Regulatory analyses during this period continue to emphasize impurity predictability, integrated control strategies, and lifecycle comparability—areas where SPOS remains structurally advantaged [10, 18].
Collectively, the recent literature supports a model of platform evolution rather than platform replacement [13, 11].
7. Timeline of Advances (2020-2025)
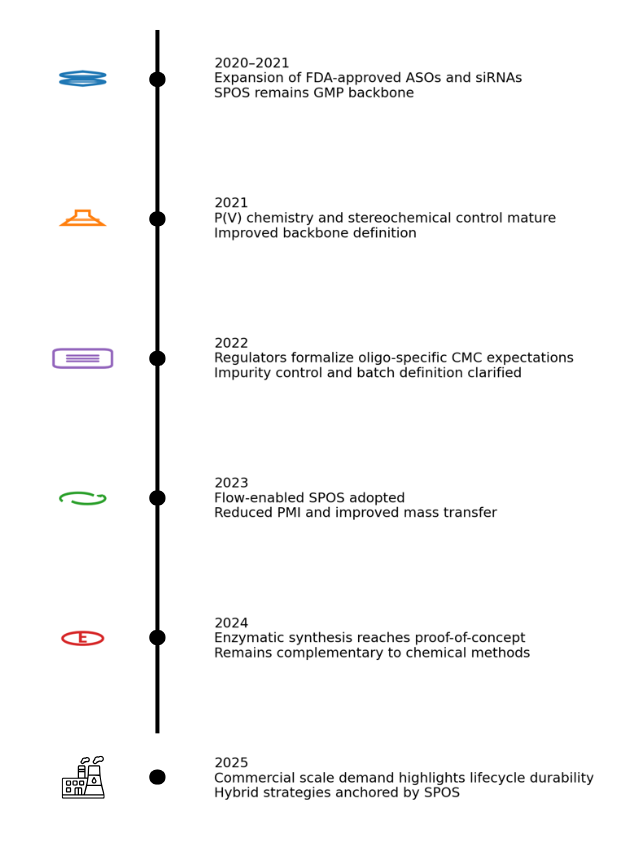
Figure 4. Timeline of key advances in oligonucleotide manufacturing (2020–2025), with icons denoting dominant technology focus per year.
8. Where Enzymatic and Hybrid Approaches Fit
Without Displacing SPOS
Recent enzymatic publications demonstrate credible progress toward controlled, template‑independent synthesis, including RNA proof‑of‑concept under aqueous conditions and reversible termination frameworks [17, 14]. At the same time, attempts to extend enzymatic synthesis into heavily modified XNA/LNA regimes highlight substrate‑tolerance and deprotection challenges—reinforcing that chemical generality remains a key limitation for enzymatic platforms in therapeutic contexts [12]. The resulting landscape is layered: SPOS (increasingly flow‑enabled) as the primary GMP backbone for chemically complex therapeutics; enzymatic synthesis for long/repetitive sequences and rapid prototyping; and hybrid strategies applied where they reduce risk rather than introduce it [13, 10].
Conclusion
The last five years have not made SPOS obsolete. They showed that SPOS can absorb innovation without losing regulatory or analytical continuity. Flow execution and greener process engineering modernize SPOS while preserving the chemistry and impurity logic regulators already understand [11, 10]. Enzymatic synthesis is accelerating—while its leading papers candidly show both momentum and remaining constraints for modified therapeutics [17, 12, 14]. The end state is convergence: SPOS remains the reference chemistry, modernized through process engineering and complemented by enzymatic and hybrid approaches where they add value without disrupting CMC [13, 10].
References
- Beaucage, S. L.; Caruthers, M. H. Deoxynucleoside phosphoramidites—A new class of key intermediates for deoxypolynucleotide synthesis. Tetrahedron Letters 1981, 22(20), 1859–1862.
- Caruthers, M. H. Gene synthesis machines: DNA chemistry and its uses. Science 1985, 230(4723), 281–285.
- Crooke, S. T. Molecular mechanisms of antisense oligonucleotides. Nucleic Acid Therapeutics 2017, 27(2), 70–77.
- Khvorova, A.; Watts, J. K. The chemical evolution of oligonucleotide therapies of clinical utility. Nature Biotechnology 2017, 35, 238–248.
- Wiles, C.; Watts, P. Continuous flow reactors: a perspective. Green Chemistry 2012, 14, 38–54.
- Jensen, K. F. Flow chemistry—Microreaction technology comes of age. AIChE Journal 2017, 63(3), 858–869.
- Palluk, S.; Arlow, D. H.; de Rond, T.; et al. De novo DNA synthesis using polymerase–nucleotide conjugates. Nature Biotechnology 2018, 36, 645–650.
- Lee, H. H.; Kalhor, R.; Goela, N.; et al. Terminator‑free template‑independent enzymatic DNA synthesis for digital information storage. Nature Communications 2019, 10, 2383.
- Roberts, T. C.; Langer, R.; Wood, M. J. A. Advances in oligonucleotide drug delivery. Nature Reviews Drug Discovery 2020, 19, 673–694.
- Wetter, C.; Chorley, C.; Curtis, C.; et al. Solution oligonucleotide APIs: regulatory considerations. Therapeutic Innovation & Regulatory Science 2022, 56, 386–393.
- Ferrazzano, L.; Corbisiero, D.; Tolomelli, A.; Cabri, W. From green innovations in oligopeptide to oligonucleotide sustainable synthesis: differences and synergies in TIDES chemistry. Green Chemistry 2023, 25, 1217–1236.
- Sabate, N.; Katkevica, D.; Pajuste, K.; et al. Towards the controlled enzymatic synthesis of LNA‑containing oligonucleotides. Frontiers in Chemistry 2023, 11, 1161462.
- Obexer, R.; Nassir, M.; Moody, E. R.; Baran, P. S.; Lovelock, S. L. Modern approaches to therapeutic oligonucleotide manufacturing. Science 2024, 384, eadl4015.
- Pichon, M.; Hollenstein, M. Controlled enzymatic synthesis of oligonucleotides. Communications Chemistry 2024, 7, 138.
- Mohammed, A. A.; AlShaer, D.; Al Musaimi, O. Oligonucleotides: evolution and innovation. Medicinal Chemistry Research 2024, 33, 2204–2220.
- Thürmer, R. EU regulators’ experience with synthetic oligonucleotides and mRNA technology. European Medicines Agency scientific conference presentation, 2023.
- Wiegand, D. J.; Rittichier, J.; Meyer, E.; et al. Template‑independent enzymatic synthesis of RNA oligonucleotides. Nature Biotechnology 2025, 43, 762–772.
- Zhang, D. (FDA CDER). Oligonucleotides: current thinking and analytical challenges identified in nusinersen PSG development (presentation), 2022.
- Al Musaimi, O.; AlShaer, D.; de la Torre, B. G.; Albericio, F. 2024 FDA TIDES (peptides and oligonucleotides) harvest. Pharmaceuticals 2025, 18(3), 291.
A CMC Philosophy for Scientists Who Think Beyond the Bench
If you’re responsible for impurity control, comparability, and lifecycle robustness—not just synthesis speed—this document is for you.
Download a clear, technical articulation of why solid‑phase synthesis continues to anchor therapeutic oligonucleotide manufacturing, and how modern flow implementations change the execution without changing the chemistry.