Difficult-To-Express Proteins Are Not Going to Solve Themselves
Difficult-To-Express Proteins Are Not Going to Solve Themselves
Innovating commercial manufacturing to meet the demand of unconventional recombinant proteins
Every now and then a researcher is tasked with finding a target recombinant protein - which ends up being a nightmare of a process. Targets such as Tissue inhibitors of metalloproteinases (TIMPs) and 6-phospho-beta-glucosidase (BGLA) are frequent candidates of this experience. These proteins fall under the dreaded list of difficult-to-express (DTE) proteins. Overcoming the complexities of DTE proteins has been an ongoing frustration - for both manufacturers and researchers. It is clear that there are multiple pieces to the DTE puzzle. There may not be a 'one size fits all' solution, but that doesn't mean DTE's are unsolvable. The main focus has always been on building and integrating tailored chemical solutions to support expression systems. My goal is to gear the focus more toward the actual expression systems and highlight their limitations from the manufacturing angle.
Problems Within a Problem
The principal issue with DTE proteins isn’t much of a mystery. The process of protein production, in one sentence, is; inserting a gene in a host cell for target protein expression(cell-free method excluded). The technical reality is that the host cell remains foreign to the target protein to some degree, regardless of conditions. The discord can be marginal enough to go unnoticed or significantly present. Consider producing a protein that is not expressed naturally in the host domain (eukaryotic, prokaryotic). This plan, expects the host to comply with potential conditions outside of its nature. In other words, it is similar to expecting a motorcycle mechanic to build a car or an auto mechanic, a bike. A degree of compatibility presents inevitable limitations.
The folding process is arguably the most condition-based step in expression. So naturally, proper confirmation is the hardest objective to achieve. It is also the priciest and most inconvenient due to its downstream position. This topic of condition compliance, however, is a door with many locks. Protein folding may be the most popular one, but it rarely acts alone. Expression difficulties are specific to target protein properties. Therefore, a more popular challenge would be overcoming varying sets of condition factors. For example, translation efficiency, codon bias, redox potential, membrane solubility, and
post-translational modifications are regular contributors to improper folding and overall expression failure.
A Solution for Solutions
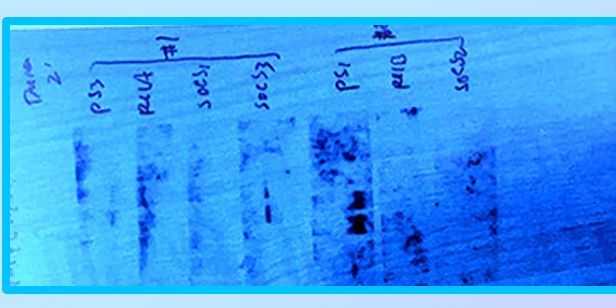
Given the many factors altering conditions, we can expect a multitude of correlating methods to develop over time. This builds an arsenal of solutions, which is not uncommon in bioresearch. However, once the list reaches a certain size, it becomes increasingly difficult to pinpoint where and which approach is the right solution for an expression. Current solutions can be sorted into the following approaches; host strain modification, manipulating gene of interest, refolding steps and chemical tweaking of conditions. Notably, the practice of utilizing tag labels has become more intuitive. Protein tags can help guide or support proteins through stages in the production process (i.e. membrane insertion, comply solubility). Their ability to measure chemistry and track key interactions has also lead to innovative screening methods. These screens have been called different names, but we know them as “expression scouts”.
Expression scouts, in short, analyze a series of varied expression systems. Their results help identify optimal factors to better achieve successful production - along with error calculation. In terms of DTE protein, this technique shows the ideal potential as a companion tool. The targeted efficiency from expression screening disempowers the role of uncalculated production errors while introducing an approach for pinpointing the right solution for an expression.
System Limitations
Pre-screening for errors is effective, but it’s not a complete approach. There is still the consideration of the standard expression systems in place. These aren’t exactly “flexible” processes. On a commercial level, expression systems set the framework for a company's manufacturing platform. Inventory, production deadlines, consumables, chemicals, and even staff expertise are all relative to these systems remaining fixed. Implementing a specialty solution for a DTE protein to them can result in a mess of outcomes. In most cases, DTE projects require their own isolated or trial expression called pilots. Even then, enduring the inflated cost and lead time can’t guarantee success. Pilots are equivalent to a company saying “sure we will try”, but their success rates aren’t traditionally comforting. The incorporation of preliminary expression scouts prior to pilots have shown rate improvements, but they aren’t dramatic. There needs to be improvements from the application process. Meaning, our standard expression systems have to become more versatile in order to meet the required changes for different protein conditions.
Suggesting “versatility” doesn’t offer any real actionable value. Not to mention, proposing such a dramatic innovation may not be received too lightly. Respectively, It is only right to acknowledge that there are a few key companies that have been working hard to build solutions. DTE proteins are just too complex to properly cover in one post. They are also too complex for just a few key companies' efforts. Not too long ago, a company required a scale of resources and funds in order to R&D such solutions. However, that excuse is starting to expire. Innovations from all angles have influenced trends in reagent manufacturing (material) cost. The price of producing protein is becoming increasingly cheaper - to the point where you would get more bang for your buck choosing custom service for > 1mg quantity than you would buying it at catalog cost. It is an opportunity for collaborative innovation. We encourage others to take advantage of this affordability and to consider implementing solutions such as expression scouts.
At the end of the day, the researcher does not need more options for the same proteins. They expect more solutions and new targets. Difficult-to-express proteins are our indirect responsibility as industry providers. They aren't going to solve themselves.